
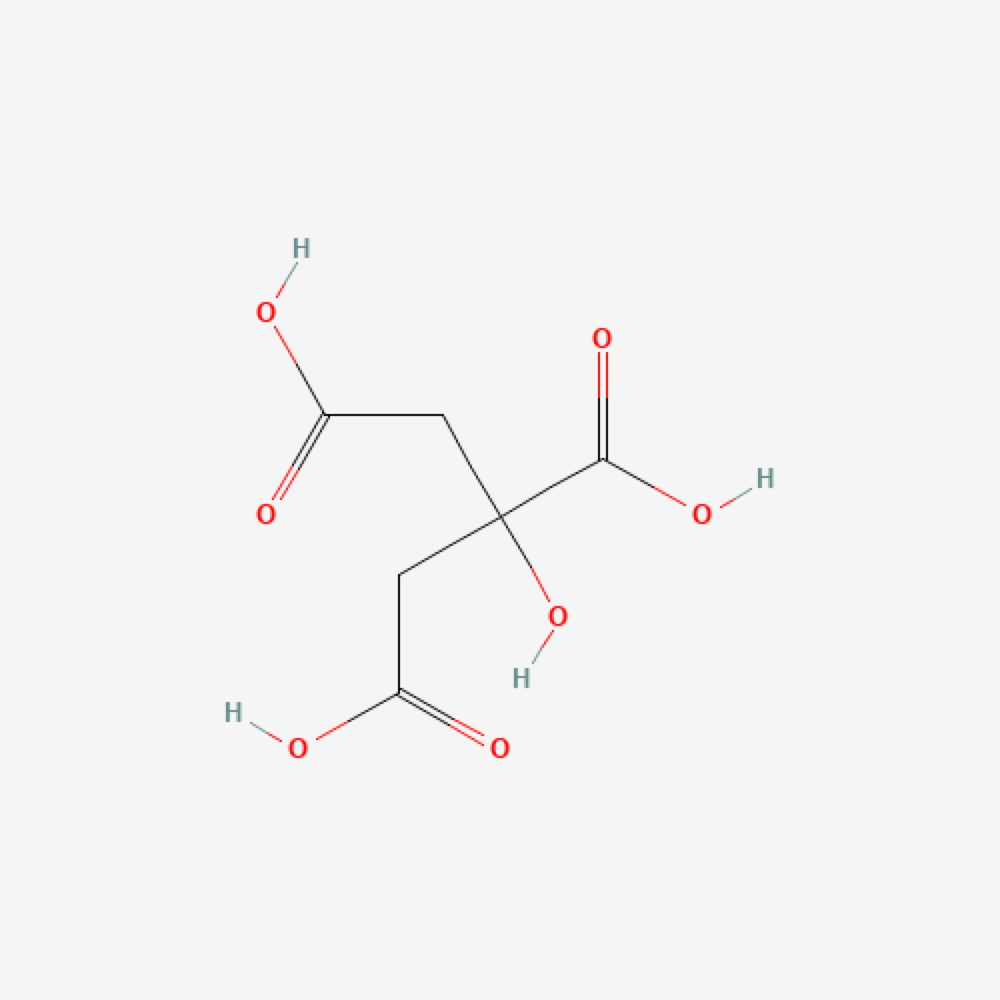
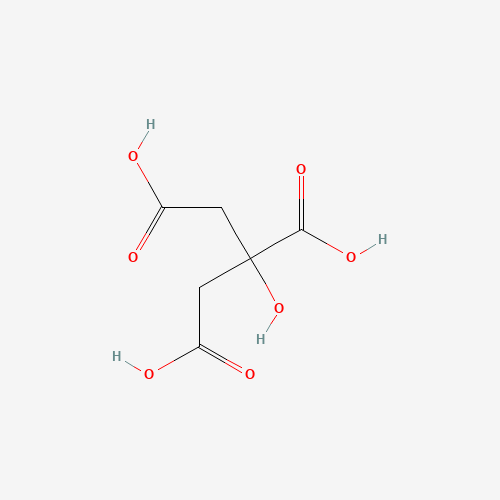

Sodium Citrate
AKA “TRISODIUM CITRATE, Citrosodine, Natrocitral, Citric acid, trisodium salt, Trisodium citrate, anhydrous, sodium citrate”
Ingredient Facts
Sodium citrate
AKA: TRISODIUM CITRATE, Citrosodine, Natrocitral, Citric acid, Trisodium salt, Trisodium citrate, Anhydrous
Synthetic · Astringent · EWG: 4
Synthetic
Humectant
Chelating Agent
Astringent
Skin Conditioner
Chemical Exfoliant
Clean
Helps balance pH and remove impurities from formulas, keeping products stable and effective on skin.
Effective at 2.5%
Solubility 29.4g/L
Formula
C6H5Na3O7
Mol. Weight
258.07
CAS #
68-04-2, 6132-04-3
Form
Powder
State
Dry Powder; Liquid; Dry Powder, Liquid, Other Solid; Other Solid; Liquid, Other Solid
See origin →
Functions
Buffering
Chelating
Grabs onto metal ions in water that would destabilize the formula. Keeps your product from going bad or changing color.
Humectant
A moisture magnet. Pulls water from the air and deeper skin layers up to the surface to keep skin plump and hydrated.
Astringent
Tightens and contracts skin tissue, temporarily shrinking pores and reducing oiliness. That "clean, tight" feeling after toner.
Antioxidant
Protects skin from free radical damage caused by UV, pollution, and stress
Moisturizing
Locks in moisture to keep skin soft and supple throughout the day
Fights Acne
Provides scent
Maintains optimal pH
Binds metal ions
Traditional Use
Citric acid from citrus fruits has been known since ancient times; sodium citrate was first synthesized in the 19th century. Used historically as a pH buffer and preservative in both food and cosmetics.
Skin Types
●all-skin-types
Best For
General
Melanin-Rich Skin
Suitable
Pregnancy & Breastfeeding Safe
Yes
EWG
4
CIR
Safe
Pregnancy
Yes
CIR Safety Findings
Not a Sensitizer
medical-approval
all-shades-safe
Concentration Guide
0.004%
5.0%
0%7%
Effective Range
CIR reported use: 0.01-2 | 0.004-5
Regulatory Agencies
Super Sculpt Serum
Alpyn Beauty
body cleansing bar
Cocokind
ceramide hydration duo
Cocokind
retinol face + body duo
Cocokind
milky-soft face and body cleanser
Cocokind
Rosilliance® Tinted Moisturizer - Fresh Rose
COOLA
Rosilliance® Tinted Moisturizer - Golden Hour
COOLA
Detox Hydrating Gel
Goldfaden MD
AM + PM Hydrating Booster Set
Goldfaden MD
Detox & Clarify Collection
Goldfaden MD
Phaze™ Gel Cleanser
Pai Skincare
Bonne Nuit™ Night Cream
Pai Skincare
Resurrection Girl™ Mask
Pai Skincare
Phaze™ Gel Cleanser Mini
Pai Skincare
Gentle Genius™ Barrier Care Wet Skin Moisturizer
Pai Skincare
Skin Renewal Serum (25mL)
Grown Alchemist
[Travel Size] Skin Renewal Serum (5mL)
Grown Alchemist
Smooth + Sculpt Set
Grown Alchemist
Tulip Dew Drops
Bloom Effects
Radiance Reset
Bloom Effects
Everyday Radiance Essentials
Bloom Effects
Balance + Support Duo
Marie Veronique
Lunar Bloom Retinal Serum
Ursa Major
Luxury Vegan Brush Soap
Jenny Patinkin
Forest Water Hyaluronic Serum
Ursa Major
Forest Fix Spray Deodorant
Ursa Major
Alpine Rich Cream
Ursa Major
Gentle Retinol Night Serum
Marie Veronique
COLORFREE ZINC Broad Spectrum Sunscreen | SPF 32
Marie Veronique
Barrier Restore Serum
Marie Veronique
Regenerating Gel Mask (20ml)
Grown Alchemist
Advanced Protection Cream
OSEA
Advanced Protection Cream - 0.25 oz
OSEA
Anti-Aging Sea Serum - 0.19 fl oz
OSEA
Water-Lock Moisturizer - 10ML
– Tata Harper Skincare
Tata Harper Skincare
Water-Lock Moisturizer
Tata Harper Skincare
Tata's Daily Essentials
Tata Harper Skincare
Superkind Softening Cleanser -15ML
– Tata Harper Skincare
Tata Harper Skincare
Superkind Softening Cleanser
Tata Harper Skincare
Superkind Refining Cleanser - 15ML
– Tata Harper Skincare
Tata Harper Skincare
Superkind Refining Cleanser
Tata Harper Skincare
Superkind Radiance Mask
Tata Harper Skincare
Superkind Essentials
Tata Harper Skincare
Resurfacing Serum - 10ML
– Tata Harper Skincare
Tata Harper Skincare
Resurfacing Body Serum - 25ml
– Tata Harper Skincare
Tata Harper Skincare
Restorative Eye Crème - 3ML
– Tata Harper Skincare
Tata Harper Skincare
Repairative Moisturizer - 10ML
– Tata Harper Skincare
Tata Harper Skincare
Repairative Moisturizer
Tata Harper Skincare
Refillable Restorative Eye Crème
Tata Harper Skincare
Raw Honey Crystal Mask
Tata Harper Skincare
Citric acid from Aspergillus niger: a comprehensive overv...
Bikash Chandra Behera
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overv...
Bikash Chandra Behera
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overv...
Bikash Chandra Behera
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overview.
Behera BC
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overview.
Behera BC
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overview.
Behera BC
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overview.
Behera BC
Critical reviews in microbiology, 2020
Citric acid from Aspergillus niger: a comprehensive overview.
Behera BC
Critical reviews in microbiology, 2020
Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics
Cosmetic Ingredient Review (CIR) Expert Panel
International Journal of Toxicology
Citric Acid
European Commission
EU Cosmetic Ingredient Database (CosIng)
Monosodium Citrate
European Commission
EU Cosmetic Ingredient Database (CosIng)
Sodium Citrate
European Commission
EU Cosmetic Ingredient Database (CosIng)
Citric Acid
Environmental Working Group (EWG)
EWG Skin Deep Cosmetics Database
Sodium Citrate
Environmental Working Group (EWG)
EWG Skin Deep Cosmetics Database
Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics.
Fiume MM, Heldreth BA, Bergfeld WF, Belsito DV, Hill RA et al.
International journal of toxicology, 2014
Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics.
Fiume MM, Heldreth BA, Bergfeld WF, Belsito DV, Hill RA et al.
International journal of toxicology, 2014
Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics.
Fiume MM, Heldreth BA, Bergfeld WF, Belsito DV, Hill RA et al.
International journal of toxicology, 2014
Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics.
Fiume MM, Heldreth BA, Bergfeld WF, Belsito DV, Hill RA et al.
International journal of toxicology, 2014
Citric acid
Health Canada
Health Canada Cosmetic Ingredient Hotlist
Verified Feb 2026
Data: PubChem · CosIng · EWG · CIR